 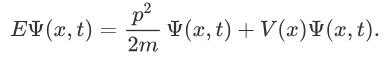
It will tell only the possible positions and probabilities of being in those possible positions. Schrodinger’s Equation doesn't tell the position of the subatomic particles at any future point in time. 
Here, in the following article, we will discuss Schrodinger’s equation in depth. In 1925, Schrodinger and Heisenberg independently synthesized the representations of quantum mechanics that successfully describe physical phenomena at the microscopic level of nuclei, molecules, and atoms. Newton's laws are deterministic because by using the given knowledge of the initial position and the measurements of the forces acting on the object, one can tell how the forces will interact, and therefore, where the object is going to be in the upcoming point of time. Although parallel, Schrodinger’s Equation is not deterministic as Newton’s laws. In classical physics, it is parallel to Newton’s Laws of Motion, which helps you to calculate the future position and momentum of the object if you know the present position and momentum of an object. Schrodinger’s Equation refers to a fundamental equation of quantum physics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
